Project C3
Doctoral researcher: Marcel Welle
Principle investigator: Christine Stöhr
Co-supervisors: F. Scholz, M. Lalk
Effect of NO on activity of root plasma membrane proteins and cell metabolites under hypoxic conditions
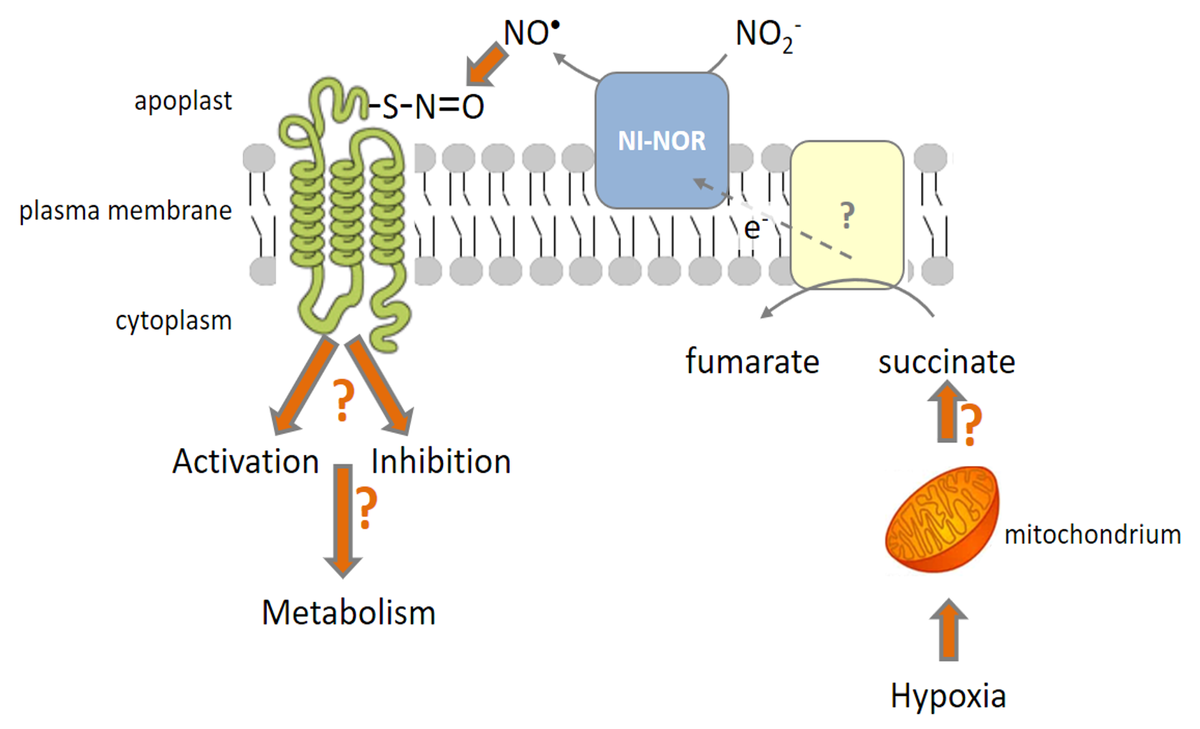
The ongoing project revealed that the formation of nitric oxide at the plasma membrane (pm) of tomato roots was highly dependent on the oxygen availability for the plant roots. In response to in vitro NO treatment, a high number of potentially S-nitrosylated membrane proteins has been located at the root plasma membrane. Beside others, several receptor-like kinases, aquaporins, H+-ATPases and redox enzymes (Cytochrome b5 reductase, glutathione-dependent disulfide oxidoreductase) were proven to react with NO. In the next period, we want to study how the NO-dependent S-nitrosylation may affect the activity of the proven pm proteins and what could be the impact on cell metabolism.
The influence of NO on pm ATPases and on activities of pm aquaporins will be in focus. A potentially NO dependent regulation of the redox capacity of root pm vesicles will be studied in tight collaboration with project B (research group of Prof. Scholz). According to the new aspects found at monolayers of disintegrated mitochondria in project B2, the voltammetric measurement with mercury electrodes will be applied to plant pm vesicles revealing changes in redox activities due to NO.
Since the plant roots form NO at the plasma membrane when subjected to hypoxic conditions (e.g. flooding), the implications on cell metabolism will be analyzed in root protoplasts. Isolated from roots under hypoxic and well-aerated conditions, the in vitro effect of NO on protoplast metabolism will be studied. Metabolic compounds will be analyzed in collaboration with Prof. Lalk with emphasis on amino acids and organic acids composition. Established GC-MS, HPLC-MS and NMR methods will be used to quantify selected metabolites.
To study and to compare the effects mediated by NO and/or hypoxia on carbon fluxes allows us to assess a supposable metabolic reconfiguration (non-circular TCA flux, alanine and 2-oxoglutarate shunt, γ-aminobutyric acid shunt; reviewed by Bailey-Serres et al. (2012)). To verify the obtained data for whole plants, the single cell analysis will be established together with Prof. Lalk (Fujii et al. (2015)). Single root cells from intact tomato plants with aerated or hypoxic roots will be probed and immediately analyzed. The impact of NO stress conditions on lipid species (like triacylglycerides, diacylglycerides and phosphatides) will be investigated using a HPLC-MS lipidomics platform in collaboration with Dr. Kristian Wende (INP Greifswald).
Literature
[1] Bailey-Serres et al. (2012) Making sense of low oxygen sensing. Trends Plant Sci. 17(3):129-38
[2] Fujii et al. (2015). Direct metabolomics for plant cells by live single-cell mass spectrometry. Nature Protocols 10, 1445–1456
Contact
Christine Stöhr
University of Greifswald
Institute of Botany and Landscape Ecology
Grimmer Strasse 88
D-17487 Greifswald
Tel: +49 (0)3834 420 4104
Fax:+49 (0)3834 86 4114
stoehruni-greifswaldde
Website